Metals like iron, steel, copper, and brass are generally not suitable for conventional anodizing.
Anodizing works best on metals that naturally form stable oxide layers, especially aluminum, titanium, and magnesium.
Non-anodizable metals may corrode, discolor, or fail to form a durable protective coating during the process.
In this article:
Anodizing only works well on certain metals that naturally form a stable, adherent oxide layer under electrochemical conditions. These are often called “valve metals.”
Many metals either:
- dissolve instead of forming oxide,
- form flaky/nonprotective oxides,
- corrode rapidly in the electrolyte,
- or cannot sustain a controlled anodic film.
Metals That Anodize Well
These metals anodize successfully:
- Aluminum
- Titanium
- Magnesium
- Niobium
- Tantalum
- Zirconium
These metals form dense oxide films.
Metals That Generally Cannot Be Properly Anodized
1. Steel (Carbon Steel)
Carbon steel
Why it fails:
- Iron oxide (“rust”) is porous and unstable.
- The oxide flakes off instead of bonding tightly.
Instead of anodizing, steel is usually protected by:
- painting
- galvanizing
- black oxide coating
- electroplating
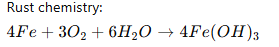
The resulting oxide is not protective like aluminum oxide.
2. Stainless Steel (Generally)
Stainless steel
Stainless steel already forms a passive chromium oxide film naturally.
Problems:
- Does not produce thick decorative anodic films like aluminum
- Conventional anodizing electrolytes do not work effectively
- Surface tends to pit or discolor
What is used instead:
- electropolishing
- passivation with nitric/citric acid
- PVD coatings
There are specialty coloring processes sometimes marketed as “stainless anodizing,” but they are not true conventional anodizing.
3. Copper
Copper
Why:
- Copper oxides are conductive and unstable.
- Oxide layer does not self-limit properly.
- Surface darkens unevenly.
Result:
- poor adhesion
- powdery oxide
- nonuniform coating
Copper is more commonly:
- plated
- chemically patinated
- lacquer coated
4. Brass
Brass
Because brass contains:
- copper
- zinc
Problems:
- selective dissolution (“dezincification”)
- uneven oxide formation
- poor coating stability
Decorative alternatives:
- lacquering
- plating
- patination
5. Bronze
Bronze
Issues:
- complex alloy chemistry
- unstable oxide mixtures
- patchy corrosion products
Usually treated with:
- waxes
- patinas
- clear coatings
6. Zinc
Zinc
Why:
- Zinc dissolves rapidly in acidic anodizing baths.
- Oxide layer is weak and chalky.
Zinc protection instead relies on:
- chromate conversion coatings
- galvanization systems
7. Tin
Tin
Problems:
- oxide layer lacks mechanical strength
- poor adhesion
- unstable film growth
Rarely anodized industrially.
8. Lead
Lead
Why:
- soft unstable oxides
- toxic electrolyte contamination
- poor film properties
Industrial use is extremely limited.
9. Nickel
Nickel
Nickel forms passive films, but:
- conventional anodizing does not create useful porous oxide
- coatings tend to crack or remain too thin
Nickel is usually:
- electroplated
- chemically passivated
10. Precious Metals
Includes:
- Gold
- Silver
- Platinum
Why they do not anodize:
- Too chemically inert
- Resist oxidation
Instead:
- polished
- plated
- coated
Why Some Metals Cannot Be Anodized
A successful anodizing metal must form an oxide that is:
| Requirement | Why Important |
|---|---|
| Adherent | Must bond tightly |
| Electrically insulating | Stops runaway corrosion |
| Chemically stable | Must resist dissolution |
| Dense and continuous | Must protect substrate |
| Self-healing/passivating | Enables controlled growth |
Many metals fail one or more of these conditions.
The “Valve Metal” Concept
True anodizable metals are usually:
- aluminum
- titanium
- tantalum
- niobium
- zirconium
These are called:
Valve metal
Their oxides behave like electrical insulators:
- current flows one way during oxide growth
- oxide thickens controllably
This is why they anodize so well.
Can Mixed Alloys Be Anodized?
Sometimes partially.
Aluminum Alloys
Aluminum alloy
Most can anodize, but alloy composition matters.
High copper alloys:
- darker finish
- lower corrosion resistance
High silicon alloys:
- gray appearance
Cast aluminum:
- uneven coloration
What Happens If You Try to Anodize the Wrong Metal?
Possible outcomes:
- rapid dissolution
- black sludge
- pitting
- gas evolution
- powdery oxides
- no coating growth
- surface burning
For example, steel in sulfuric anodizing solution may simply corrode aggressively instead of forming a useful coating.
Related Processes Used Instead of Anodizing
| Metal | Alternative Process |
|---|---|
| Steel | Galvanizing, black oxide |
| Stainless steel | Passivation, electropolishing |
| Copper/brass | Patination, lacquer |
| Zinc | Chromate conversion |
| Nickel | Electroplating |
| Gold/silver | Polishing/plating |
Important Distinction
Some metals can undergo:
- electrochemical oxidation
without producing a useful industrial anodized coating.
So “can oxidize electrically” is not the same as:
- “can be commercially anodized successfully.”
Summary
Metals commonly NOT suitable for conventional anodizing:
- carbon steel
- stainless steel
- copper
- brass
- bronze
- zinc
- tin
- lead
- nickel
- gold
- silver
- platinum
Metals ideal for anodizing:
- aluminum
- titanium
- magnesium
- niobium
- tantalum
- zirconium
The key factor is whether the metal forms a stable, dense, insulating oxide layer during electrochemical oxidation.
Other courses:



