Anodizing mainly uses sulfuric acid as the electrolyte chemical to create a protective oxide layer on aluminum.
Other chemicals like chromic acid, oxalic acid, and sea ling agents may also be used depending on the finish and application.
The process improves corrosion resistance, durability, and surface appearance of metals.
In this article:
Chemicals Used in Anodizing
Anodizing is an electrochemical process that thickens the natural oxide layer on metals—most commonly aluminum—to improve corrosion resistance, wear resistance, dye adhesion, and appearance. The main “chemical used in anodizing” depends on the anodizing type and the stage of the process.
The most common anodizing electrolyte is:
Sulfuric acid
- Chemical formula: H₂SO₄
- Used in: Type II sulfuric anodizing (the industry standard)
- Concentration: typically 10–20% by volume
- Function:
- Conducts electricity
- Dissolves part of the oxide while the coating grows
- Helps create a porous aluminum oxide layer
This process forms:
- Aluminum oxide (Al₂O₃)
Main Chemicals Used Across the Full Anodizing Process
Anodizing is actually a sequence of chemical treatments.
1. Cleaning / Degreasing
Before anodizing, oils and contaminants are removed.
Common chemicals:
- Sodium hydroxide (NaOH)
- Surfactants
- Detergent alkaline cleaners
Purpose:
- Remove grease, machining oils, fingerprints
- Prepare uniform surface
2. Etching
Used to matte or texture aluminum.
Main chemical:
- Sodium hydroxide
Reaction:
- Dissolves some aluminum surface
- Produces a satin finish
Important:
- Excess etching can damage dimensional accuracy.
3. Desmutting / Deoxidizing
After etching, alloying residues (“smut”) remain.
Common chemicals:
- Nitric acid
- Ferric sulfate
- Ammonium bifluoride
Purpose:
- Remove copper, silicon, and alloy residues
- Produce a clean reactive surface
Main Anodizing Electrolytes
A) Sulfuric Acid Anodizing (Most Common)
Sulfuric acid
Characteristics:
- Produces clear porous oxide
- Good for dyeing
- Economical
- Widely used in:
- consumer electronics
- architectural aluminum
- automotive parts
Typical conditions:
- 15–22°C
- DC current
- 12–24 V
Coating thickness:
- ~5–25 microns
B) Hard Anodizing
Also uses:
Sulfuric acid
But:
- Lower temperatures (0–5°C)
- Higher current density
Result:
- Much harder coating
- Thick wear-resistant oxide
Applications:
- aerospace
- hydraulic cylinders
- firearm parts
- industrial tooling
Typical hardness:
- Comparable to hardened steel in wear resistance
C) Chromic Acid Anodizing
Chromic acid
- Formula: H₂CrO₄
Advantages:
- Thin but corrosion-resistant coating
- Good for aerospace fatigue-sensitive parts
Disadvantages:
- Toxic
- Contains hexavalent chromium
Because of environmental concerns, it is increasingly replaced.
Related hazard:
- Cancer risk from Cr⁶⁺ compounds
D) Phosphoric Acid Anodizing
Phosphoric acid
Purpose:
- Creates large pores
- Excellent for adhesive bonding
Used in:
- aircraft structural bonding
E) Oxalic Acid Anodizing
Oxalic acid
Characteristics:
- Hard decorative finish
- Sometimes yellowish tint
More common historically in Europe and Japan.
Dyeing Chemicals
After sulfuric anodizing, the oxide is porous and can absorb dyes.
Used chemicals:
- Organic dyes
- Metal salt dyes
Common colors:
- black
- bronze
- blue
- red
- gold
Electrolytic coloring may use:
- tin salts
- cobalt salts
- nickel salts
Sealing Chemicals
After anodizing, pores are sealed to improve corrosion resistance.
Hot Water Sealing
Uses:
- deionized boiling water
Converts oxide into hydrated form.
Nickel Acetate Sealing
Nickel acetate
Advantages:
- Better corrosion resistance
- Better color retention
Electrochemical Principle
The aluminum part acts as the:
- Anode (positive electrode)
Hence the term:
- “anodizing”
At the anode:
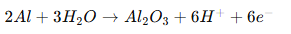
The oxide grows inward and outward simultaneously.
Metals That Can Be Anodized
Besides aluminum:
- Titanium
- Magnesium
- Niobium
Titanium anodizing often uses:
- phosphoric acid
- sulfuric acid
- trisodium phosphate solutions
Color is created by optical interference rather than dyes.
Safety Considerations
Many anodizing chemicals are hazardous:
- strong acids
- caustic alkalis
- fluoride compounds
- chromium compounds
Risks include:
- chemical burns
- toxic fumes
- environmental contamination
Industrial anodizing requires:
- ventilation
- PPE
- wastewater treatment
- electrical safety controls
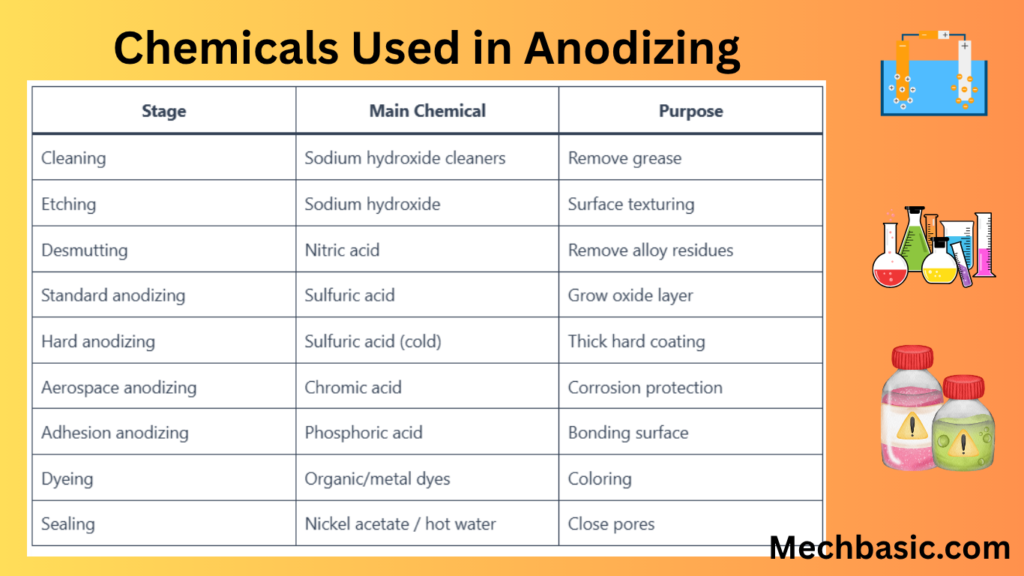
Summary Table
| Stage | Main Chemical | Purpose |
|---|---|---|
| Cleaning | Sodium hydroxide cleaners | Remove grease |
| Etching | Sodium hydroxide | Surface texturing |
| Desmutting | Nitric acid | Remove alloy residues |
| Standard anodizing | Sulfuric acid | Grow oxide layer |
| Hard anodizing | Sulfuric acid (cold) | Thick hard coating |
| Aerospace anodizing | Chromic acid | Corrosion protection |
| Adhesion anodizing | Phosphoric acid | Bonding surface |
| Dyeing | Organic/metal dyes | Coloring |
| Sealing | Nickel acetate / hot water | Close pores |
Other courses:



