Anodizing is an electrochemical process that thickens the natural oxide layer on a metal surface, mainly aluminum.
The metal is immersed in an acid electrolyte bath and electric current is passed through it to form the coating.
This process improves corrosion resistance, hardness, durability, and decorative appearance.
In this article:
- Anodizing Process
- Anodizing-Definition:
- Basic Principle
- History of Anodizing
- Metals That Can Be Anodized
- Main Components of an Anodizing Setup
- How the Anodizing Process Works
- Types of Anodizing
- Important Process Parameters
- Advantages of Anodizing
- Disadvantages of Anodizing
- Applications of Anodizing
- Difference Between Anodizing and Electroplating
- Summary
Anodizing Process
What Is Anodizing?
Anodizing is an electrochemical surface treatment process used mainly to increase the thickness of the natural oxide layer on metals, especially:
- Aluminum
- Titanium
- Magnesium
The process improves:
- Corrosion resistance
- Surface hardness
- Wear resistance
- Appearance
- Paint adhesion
- Electrical insulation
Unlike electroplating, anodizing does not deposit another metal on the surface. Instead, it converts the metal surface itself into a durable oxide layer.
Anodizing-Definition:
The metal object being treated becomes the anode in an electrolytic cell.
That is why the process is called anodizing.
Basic Principle
When electric current passes through an electrolyte solution, oxygen reacts with the metal surface to form a thick oxide coating.
For aluminum:
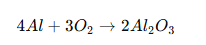
The oxide formed is:
- Hard
- Porous
- Strongly attached to the metal
- Corrosion resistant
History of Anodizing
Modern anodizing developed in the 1920s for protecting seaplane parts from corrosion.
Major industrial growth occurred in:
- Aerospace
- Architecture
- Automotive industries
Today anodized aluminum is widely used in:
- Mobile phones
- Kitchen utensils
- Window frames
- Aircraft parts
- Electronics
Metals That Can Be Anodized
Most Common
Aluminum
The most widely anodized metal because it forms a stable oxide layer.
Titanium
Used for:
- Medical implants
- Aerospace parts
- Colored decorative finishes
Magnesium
Used in lightweight engineering components.
Main Components of an Anodizing Setup
An anodizing system contains:
| Component | Function |
|---|---|
| Anode | Metal object to be anodized |
| Cathode | Usually lead, stainless steel, or aluminum |
| Electrolyte | Acid solution |
| DC Power Supply | Provides current |
| Tank | Holds electrolyte |
How the Anodizing Process Works
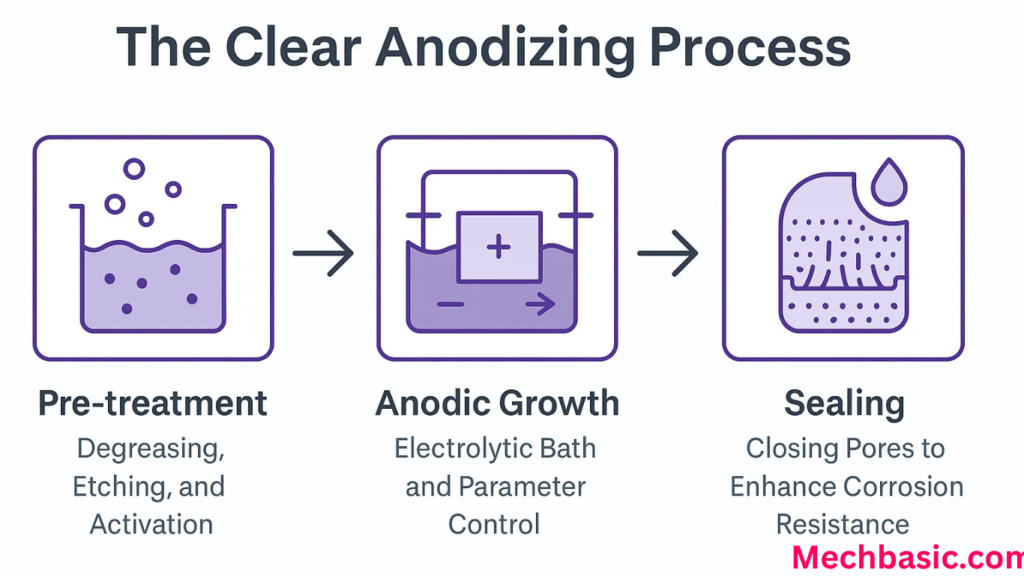
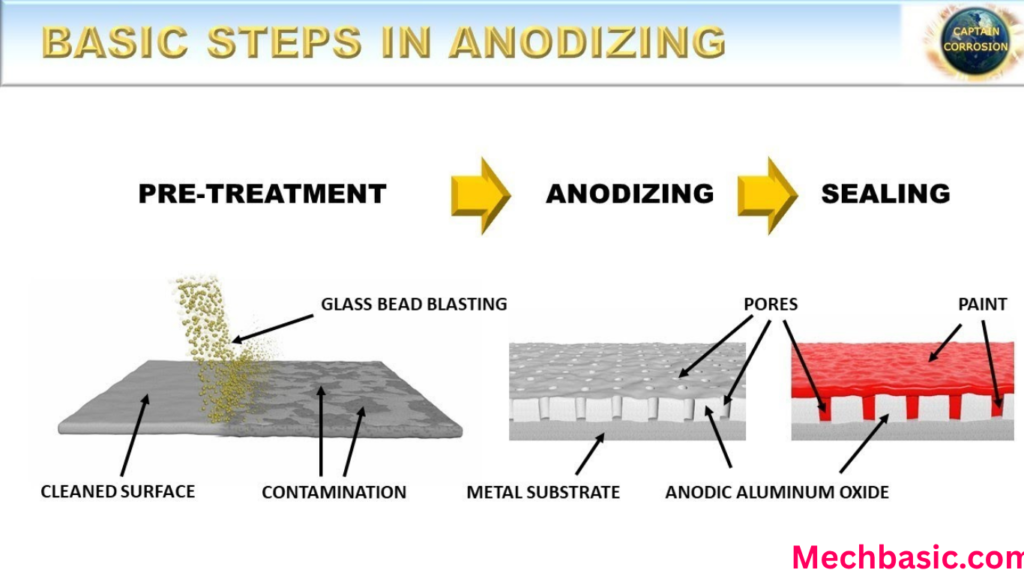
Step 1: Cleaning
The metal surface is cleaned to remove:
- Oil
- Dirt
- Grease
- Oxides
Methods:
- Degreasing
- Chemical cleaning
- Rinsing
Step 2: Etching
The metal is chemically etched to create a uniform surface finish.
For aluminum, sodium hydroxide is commonly used.
Step 3: Desmutting
Removes impurities left after etching.
Nitric acid is often used.
Step 4: Anodizing
The metal is placed in an acid electrolyte bath and connected to the positive terminal.
The cathode is connected to the negative terminal.
Common electrolyte:
- Sulfuric acid
The electrochemical reaction:
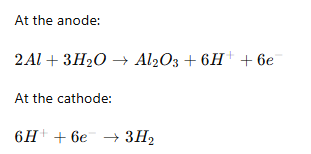
Result:
- Oxygen combines with aluminum
- Oxide layer grows on the surface
Structure of the Anodized Layer
The anodized coating has two parts:
Barrier Layer
- Thin
- Dense
- Non-porous
Porous Layer
- Thicker
- Contains microscopic pores
- Can absorb dyes and sealants
Step 5: Coloring (Optional)
The porous oxide layer can absorb dyes.
Common colors:
- Black
- Blue
- Red
- Gold
- Bronze
Methods:
- Dye coloring
- Electrolytic coloring
- Integral coloring
Step 6: Sealing
Sealing closes the pores to improve:
- Corrosion resistance
- Durability
- Color retention
Common sealing methods:
- Hot water sealing
- Steam sealing
- Nickel acetate sealing
Hydration reaction:
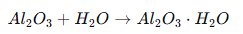
Types of Anodizing
Type I — Chromic Acid Anodizing
Uses:
- Aerospace parts
Characteristics:
- Thin coating
- Good corrosion resistance
Type II — Sulfuric Acid Anodizing
Most common type.
Features:
- Decorative finish
- Can be dyed
- Moderate hardness
Type III — Hard Anodizing (Hardcoat)
Uses:
- Machine parts
- Pistons
- Cylinders
Characteristics:
- Very thick
- Extremely hard
- Wear resistant
Important Process Parameters
| Parameter | Effect |
|---|---|
| Voltage | Controls coating growth |
| Current density | Influences coating thickness |
| Temperature | Affects pore size and hardness |
| Time | Determines oxide thickness |
| Electrolyte concentration | Controls reaction speed |
Advantages of Anodizing
Excellent Corrosion Resistance
Protects metal from oxidation and weathering.
Increased Hardness
Hard anodized surfaces can approach ceramic hardness.
Attractive Appearance
Provides decorative finishes and colors.
Better Paint Adhesion
Porous surface bonds well with paints and adhesives.
Electrical Insulation
Oxide layer is non-conductive.
Environmentally Safer
Compared with some plating methods.
Disadvantages of Anodizing
- Limited mainly to aluminum and similar metals
- Coating can crack under severe deformation
- Some processes use hazardous acids
- Color matching can be difficult
Applications of Anodizing
Aerospace
Aircraft structural parts
Automotive
Engine components, wheels
Electronics
Phone bodies, laptop casings
Architecture
Doors, windows, curtain walls
Medical
Titanium implants
Consumer Goods
Cookware, sports equipment
Difference Between Anodizing and Electroplating
| Feature | Anodizing | Electroplating |
|---|---|---|
| Process | Forms oxide layer | Deposits another metal |
| Surface | Part of base metal | Separate metal coating |
| Main Metals | Aluminum, titanium | Many metals |
| Conductivity | Insulating | Usually conductive |
| Durability | Very durable | Depends on coating |
Example: Aluminum Anodizing
Industrial anodizing of aluminum commonly uses:
- Sulfuric acid electrolyte
- Voltage: 12–24 V
- Temperature: 18–22°C
Typical oxide thickness:
- Decorative: 5–25 µm
- Hard anodizing: 25–150 µm
Summary
Anodizing is an electrochemical process in which a metal—usually aluminum—is made the anode in an electrolytic cell to produce a hard, protective oxide layer on its surface.
The process:
- Cleans the metal
- Oxidizes the surface electrically
- Produces a porous oxide layer
- Allows coloring and sealing
It is widely used because it improves:
- Corrosion resistance
- Hardness
- Appearance
- Durability
without adding a separate coating material.
Other courses:



