Electroplating was developed in the early 19th century by scientists experimenting with electricity and metal deposition.
Luigi Brugnatelli is credited with inventing modern electroplating in 1805 using electrochemical methods.
His work laid the foundation for industrial electroplating used in manufacturing, jewelry, and engineering today.

In this article:
Who Invented Electroplating?
Electroplating developed gradually through the work of several scientists during the late 18th and early 19th centuries. There is no single universally accepted “sole inventor,” but the modern electroplating process is mainly credited to:
Luigi Brugnatelli
He is widely regarded as the first person to successfully perform electroplating using electricity.
The invention of electroplating is closely connected to the early development of electricity and electrochemistry in the 18th and 19th centuries. No single person “invented” it all at once; instead, several scientists and inventors contributed key discoveries that led to modern electroplating.
What Is Electroplating?
Electroplating is the process of coating one metal with a thin layer of another metal using electricity. It is commonly used for:
- Preventing corrosion
- Improving appearance
- Increasing hardness
- Enhancing conductivity
Examples include gold-plated jewelry, chrome-plated car parts, and silver-plated utensils.
The basic principle is:
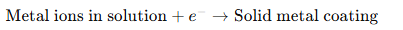
Early Scientific Foundations
Luigi Galvani (1737–1798)
Galvani discovered that electricity could cause chemical and biological reactions. His experiments with frog legs in the 1780s helped establish the relationship between electricity and chemistry.
Although he did not invent electroplating, his work inspired later electrochemists.
Alessandro Volta (1745–1827)
Volta invented the electric battery in 1800:
Voltaic Pile
This was the first continuous source of electric current. Without a steady electric current, electroplating would not have been possible.
The invention of the battery made practical electrochemical experiments possible.
Discovery of Electroplating
Luigi Valentino Brugnatelli — The First Electroplater
Most historians credit Brugnatelli with inventing electroplating in 1805.
Using Volta’s battery, Brugnatelli successfully coated objects with a thin layer of gold through electrochemical deposition.
His process involved passing electric current through a solution containing dissolved gold.
This reaction can be represented as:
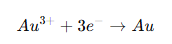
That means gold ions gained electrons and became solid gold on the surface of another object.
Why His Work Was Not Immediately Famous
Brugnatelli had disagreements with the scientific establishment, especially with Volta and the French scientific community under Napoleon Bonaparte. Because of this, his work received little publicity and was largely ignored for many years.
So although he demonstrated electroplating first, the technology did not spread widely at that time.
Industrial Development of Electroplating
George Richards Elkington and Henry Elkington
In the 1830s and 1840s, the Elkingtons transformed electroplating from a laboratory experiment into a commercial industry in United Kingdom.
They patented improved electroplating methods, especially for silver and gold plating.
A major breakthrough came when they used cyanide-based electrolyte solutions, which produced smoother and more durable coatings.
In 1840, they received important patents that allowed electroplating to become commercially successful.
Their company became one of the world’s leading electroplating manufacturers.
Important Scientific Contributions
Michael Faraday
Faraday did not invent electroplating, but he explained the scientific laws behind it.
His laws of electrolysis showed how the amount of deposited metal depends on electric current and time.
One simplified form is:
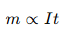
where:
- (m) = mass deposited
- (I) = current
- (t) = time
These laws became fundamental to industrial electroplating.
How Electroplating Works
A typical electroplating setup contains:
- Cathode — the object to be plated
- Anode — the plating metal
- Electrolyte solution — contains metal ions
- Power supply — provides electric current
Example with copper plating:
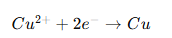
Copper ions gain electrons and form a solid copper layer on the object.
Timeline of Electroplating History
| Year | Event |
|---|---|
| 1800 | Volta invents the battery |
| 1805 | Brugnatelli performs first electroplating |
| 1830s | Electrochemistry advances rapidly |
| 1840 | Elkingtons patent industrial electroplating |
| Late 1800s | Electroplating widely used in manufacturing |
Modern Uses of Electroplating
Today electroplating is used in:
- Electronics
- Aerospace
- Jewelry
- Automotive manufacturing
- Medical equipment
- Coins and cutlery
Common plating metals include:
- Gold
- Silver
- Nickel
- Chromium
- Copper
- Zinc
Conclusion
The person most widely credited with inventing electroplating is Luigi Valentino Brugnatelli in 1805. He used electricity from Alessandro Volta’s battery to deposit gold onto metal surfaces.
However, modern electroplating became commercially practical later through the industrial work of George Richards Elkington and Henry Elkington in the 1840s.
Other courses:



