Some metals like titanium, tungsten, and niobium are difficult to electroplate because they form stable oxide layers on their surfaces.
Non-conductive materials such as plastics, rubber, and ceramics also cannot be directly electroplated without special surface treatment.
Proper surface preparation and activation are often needed to improve plating on hard-to-plate metals.
In this article:
Metals That Cannot Be Easily Electroplated
Electroplating works best when a metal surface:
- Conducts electricity well
- Is chemically stable in plating solutions
- Allows strong adhesion of deposited metal
- Does not instantly form insulating oxide layers
Some metals are very difficult, impractical, or nearly impossible to electroplate directly under normal conditions.
Categories of Metals That Cannot Be Easily Electroplated
These metals usually fall into one of the following categories:
| Problem | Effect |
|---|---|
| Rapid oxide formation | Prevents adhesion |
| Poor conductivity | Stops uniform current flow |
| High chemical reactivity | Causes unwanted reactions |
| Passive surface layers | Blocks deposition |
| Dissolution in electrolyte | Surface gets damaged |
| Hydrogen embrittlement sensitivity | Coating failure |
1. Reactive Metals
Highly reactive metals oxidize immediately when exposed to air or water.
Their oxide films prevent plating adhesion.
Aluminum
Aluminum is difficult to electroplate directly because it instantly forms a very stable oxide layer:
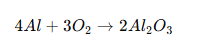
The oxide layer:
- Forms within milliseconds
- Is electrically insulating
- Prevents deposited metal from bonding
Can Aluminum Be Plated?
Yes, but only after special pretreatment such as:
- Zincate process
- Double zincate treatment
- Electroless nickel strike
Without these treatments, plating peels off easily.
Magnesium
Magnesium is even more difficult than aluminum because it:
- Corrodes rapidly
- Reacts strongly with acids
- Forms unstable oxide films
Special conversion coatings are required before plating.
Applications:
- Aerospace components
- Lightweight automotive parts
Titanium
Titanium forms an extremely stable oxide coating:
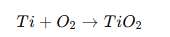
This oxide:
- Prevents metal adhesion
- Is chemically passive
- Is highly corrosion resistant
Special Methods Needed
Titanium usually requires:
- Acid activation
- Vacuum deposition
- Electroless plating
- Plasma treatments
before electroplating can succeed.
2. Metals with Passive Surfaces
Some metals naturally become “passive,” meaning they develop protective films that block electrochemical reactions.
Chromium
Chromium is difficult to plate onto directly because:
- It passivates rapidly
- Surface oxides interfere with bonding
Ironically, chromium itself is widely used AS a plating metal.
Direct chromium-on-chromium plating is difficult without activation.
Stainless Steel
Stainless steel contains chromium, which creates a passive chromium oxide film.
This film prevents good adhesion.
Pretreatments include:
- Acid etching
- Nickel strike coating
- Electrocleaning
3. Very Active Alkali and Alkaline Earth Metals
These metals cannot be electroplated from aqueous solutions because they react violently with water.
Sodium
Potassium
Calcium
These metals are impossible to electroplate from water-based electrolytes because water decomposes first:
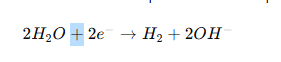
Hydrogen gas forms before the metal ions can deposit.
Result
Instead of plating:
- Water breaks down
- Hydrogen evolves
- Metal deposition fails
These metals require:
- Molten salt electrolysis
- Non-aqueous electrolytes
- Vacuum techniques
4. Metals with Extremely Negative Electrode Potentials
Metals that strongly prefer staying as ions are difficult to deposit from normal solutions.
Examples:
- Lithium
- Cesium
- Rubidium
These cannot be electroplated in ordinary aqueous baths.
5. Brittle or Hydrogen-Sensitive Metals
Some metals suffer damage during electroplating because hydrogen enters the metal.
High-strength steel
Electroplating may cause:
Hydrogen Embrittlement
Hydrogen atoms diffuse into the metal and make it brittle.
Reaction:
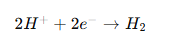
This is especially dangerous in:
- Aircraft parts
- Bolts
- Springs
Special baking treatments are required after plating.
6. Non-Conductive “Metals” or Metal Oxides
Some metallic materials lose conductivity because of thick oxide formation.
Examples:
- Heavily oxidized surfaces
- Ceramic-metal composites
These require conductive activation before plating.
Why Some Metals Are Hard to Electroplate
The success of electroplating depends on electrode potential.
A metal ion can deposit only if reduction is energetically favorable.
General deposition reaction:
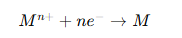
If another reaction happens more easily (such as hydrogen evolution), plating fails.
Metals That Are Easy to Electroplate
For comparison, these metals plate easily:
| Easy Metals | Reason |
|---|---|
| Copper | Excellent conductivity |
| Nickel | Good adhesion |
| Silver | Stable deposition |
| Gold | Chemically stable |
| Zinc | Easy electrochemistry |
| Tin | Low deposition difficulty |
Can “Unplatable” Metals Still Be Coated?
Yes. Modern industry uses special methods.
Alternative Methods
Electroless Plating
Uses chemical reduction instead of electricity.
Useful for:
- Aluminum
- Plastics
- Titanium
Vacuum Deposition
Examples:
- PVD
- CVD
Used for:
- Titanium nitride coatings
- Semiconductor industries
Plasma Spraying
Sprays molten metal onto surfaces.
Anodizing
Used mainly for aluminum and titanium instead of electroplating.
Practical Industrial Examples
| Metal | Difficulty | Typical Solution |
|---|---|---|
| Aluminum | Oxide layer | Zincate pretreatment |
| Magnesium | Corrosion | Conversion coating |
| Titanium | Passive oxide | Vacuum coating |
| Sodium | Water reactivity | Molten salt process |
| Stainless steel | Passivation | Nickel strike |
| High-strength steel | Hydrogen embrittlement | Post-bake treatment |
Summary
Metals cannot be electroplated easily when they:
- Form insulating oxide layers rapidly
- React strongly with water
- Become chemically passive
- Prefer remaining as ions
- Suffer hydrogen damage
The most difficult metals include:
- Aluminum
- Magnesium
- Titanium
- Sodium
- Potassium
- Calcium
However, with special pretreatments and advanced technologies, even many “difficult” metals can now be coated successfully in modern industries.
Other courses:



