High-quality electroplating requires proper surface cleaning, correct bath chemistry, and controlled temperature and current settings.
Using pure chemicals, clean equipment, and good agitation helps produce smooth and uniform coatings.
Regular testing and maintenance of the plating bath ensure strong adhesion, durability, and consistent finish quality.
In this article:
- How to Achieve High-Quality Electroplating
- “High-Quality Electroplating”-Definition:
- 1. Perfect Surface Preparation (Most Important Step)
- 2. Use High-Purity Chemicals
- 3. Maintain Correct Bath Chemistry
- 4. Correct Current Density
- 5. Proper Temperature Control
- 6. Proper Agitation
- 7. Excellent Filtration
- 8. Correct Anode Selection
- 9. Use Proper Rack and Fixture Design
- 10. Prevent Contamination
- 11. Control Deposit Thickness
- 12. Prevent Hydrogen Embrittlement
- 13. Use Correct Pretreatment for Aluminum
- 14. Optimize Additives
- 15. Good Rinsing Practice
- 16. Post-Treatment Improves Quality
- 17. Control Internal Stress
- 18. Maintain Clean Working Environment
- 19. Regular Testing and Inspection
- 20. Operator Skill and Process Discipline
- Summary
How to Achieve High-Quality Electroplating
High-quality electroplating requires precise control of:
- surface preparation,
- bath chemistry,
- electrical parameters,
- temperature,
- agitation,
- rinsing,
- filtration,
- and post-treatment.
Excellent plating is not produced by the plating bath alone — it depends on the entire process chain.
“High-Quality Electroplating”-Definition:
A good plated coating should have:
| Property | Desired Result |
|---|---|
| Adhesion | Strong metal bonding |
| Uniformity | Even thickness |
| Appearance | Bright/smooth if decorative |
| Corrosion resistance | Long-lasting protection |
| Hardness | Suitable wear resistance |
| Low porosity | Minimal defects |
| Low stress | No cracking/peeling |
| Correct thickness | Meets specification |
1. Perfect Surface Preparation (Most Important Step)
Many plating failures originate here.
Why Surface Preparation Matters
Electroplating relies on:
Atomic bonding
Contaminants block metal-to-metal bonding.
Even microscopic oil films can ruin adhesion.
Surface Preparation Sequence
Typical sequence:
Degreasing
↓
Alkaline Cleaning
↓
Water Rinse
↓
Acid Activation
↓
Water Rinse
↓
Electroplating
A) Degreasing
Removes:
- oils,
- grease,
- wax,
- machining fluid.
Methods:
- solvent cleaning
- vapor degreasing
- ultrasonic cleaning
Common chemicals:
- Sodium hydroxide
- surfactants
B) Mechanical Cleaning
Removes:
- oxides,
- rust,
- scratches.
Methods:
- polishing
- buffing
- blasting
Surface finish before plating strongly affects final appearance.
C) Acid Activation
Light acid etching removes oxide films.
Examples:
- Hydrochloric acid
- Sulfuric acid
This exposes fresh active metal.
2. Use High-Purity Chemicals
Impurities ruin plating quality.
Common High-Quality Bath Components
Nickel Plating Example
Main chemicals:
- Nickel sulfate
- Nickel chloride
- Boric acid
Why Purity Matters
Contaminants can cause:
- pitting,
- roughness,
- discoloration,
- brittleness.
Especially harmful:
- iron contamination,
- organic breakdown products,
- suspended solids.
3. Maintain Correct Bath Chemistry
The plating bath must remain within specification.
Critical variables:
| Parameter | Importance |
|---|---|
| Metal concentration | Deposition rate |
| pH | Deposit quality |
| Conductivity | Current flow |
| Additives | Brightness/leveling |
| Contamination level | Defect prevention |
pH Control
Incorrect pH causes:
- burning,
- dull deposits,
- precipitation,
- poor adhesion.
Example precipitation:
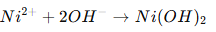
This creates rough sludge.
Monitoring Methods
Use:
- titration,
- Hull cell tests,
- spectroscopy,
- conductivity measurement.
4. Correct Current Density
One of the most critical variables.
Current density:
- current per surface area
Measured in:
- A/dm²
Too High Current
Causes:
- burning,
- roughness,
- dark edges,
- dendrites.
Too Low Current
Causes:
- dull finish,
- poor coverage,
- slow deposition.
Why Current Matters
Electroplating follows:
Faraday’s laws of electrolysis
Metal deposition amount depends on current and time.
Current Distribution Problems
Edges receive higher current density.
This may cause:
- edge burning,
- uneven thickness.
Solutions
Use:
- shields,
- auxiliary anodes,
- robbers,
- optimized rack design.
5. Proper Temperature Control
Temperature affects:
- deposition rate,
- crystal structure,
- stress,
- brightness.
Example
Nickel plating often operates around:
- 50–60°C
Hard chromium plating is highly temperature sensitive.
Using:
Chromic acid
small temperature shifts can affect:
- hardness,
- crack structure,
- efficiency.
Too Hot
Causes:
- additive decomposition,
- soft deposits,
- poor brightness.
Too Cold
Causes:
- low conductivity,
- slow plating,
- dull appearance.
6. Proper Agitation
Agitation improves:
- ion transport,
- temperature uniformity,
- deposit smoothness.
Without Agitation
Problems:
- pitting,
- burning,
- uneven thickness,
- dull deposits.
Methods
- air agitation
- solution pumping
- cathode movement
7. Excellent Filtration
Continuous filtration removes:
- particles,
- sludge,
- contaminants.
Poor Filtration Causes
- nodules,
- pits,
- rough plating,
- embedded particles.
Typical Systems
- cartridge filters
- activated carbon treatment
- continuous circulation
8. Correct Anode Selection
The anode affects:
- metal replenishment,
- bath stability.
Common Anodes
| Plating | Typical Anode |
|---|---|
| Nickel | Nickel anode |
| Copper | Copper anode |
| Chromium | Lead alloy anode |
Poor Anode Maintenance Causes
- sludge,
- contamination,
- unstable chemistry.
9. Use Proper Rack and Fixture Design
Bad fixturing causes:
- uneven current flow,
- poor electrical contact,
- shadow areas.
Good Rack Design
Should provide:
- low resistance,
- uniform exposure,
- strong electrical contact.
10. Prevent Contamination
Contamination is a major quality killer.
Sources
- dirty parts,
- drag-in,
- impure water,
- corroded equipment,
- airborne dust.
Prevention
Use:
- deionized water,
- dedicated rinse tanks,
- tank covers,
- filtration.
11. Control Deposit Thickness
Thickness must match application.
Too Thin
Problems:
- corrosion failure,
- poor wear resistance.
Too Thick
Problems:
- stress cracking,
- peeling,
- dimensional issues.
Thickness Control
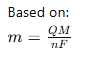
This is derived from:Faraday’s laws of electrolysis
12. Prevent Hydrogen Embrittlement
Critical for high-strength steel.
Hydrogen absorbed during plating can cause:
- cracking,
- delayed failure.
Prevention
Post-plating bake:
- 190–230°C
Especially important after:
- zinc plating,
- cadmium plating.
13. Use Correct Pretreatment for Aluminum
Aluminum naturally forms oxide:
Passivation
This blocks plating adhesion.
Solution
Use:
- zincate treatment,
- double zincate process.
14. Optimize Additives
Additives improve:
- brightness,
- leveling,
- grain refinement.
Examples:
- brighteners
- wetting agents
- stress reducers
Too Much Additive Causes
- brittleness,
- haze,
- poor adhesion.
15. Good Rinsing Practice
Proper rinsing prevents:
- contamination,
- stains,
- chemical carryover.
Best Practices
- multiple rinses
- counterflow rinsing
- spray rinses
- conductivity monitoring
16. Post-Treatment Improves Quality
After plating:
- passivation,
- sealing,
- polishing,
- baking,
- chromating
may be used.
Example
Zinc plating often followed by:
Chromate conversion coating
17. Control Internal Stress
High internal stress causes:
- cracking,
- peeling,
- warping.
Controlled using:
- additives,
- temperature,
- current density optimization.
18. Maintain Clean Working Environment
Dust can create:
- pits,
- embedded particles,
- cosmetic defects.
High-end decorative plating often uses:
- filtered air,
- controlled environments.
19. Regular Testing and Inspection
Professional plating operations continuously test:
- thickness,
- adhesion,
- hardness,
- corrosion resistance,
- porosity.
Common Test Methods
| Test | Purpose |
|---|---|
| XRF | Thickness |
| Salt spray | Corrosion resistance |
| Bend test | Adhesion |
| Microscopy | Defect analysis |
| Hull cell | Bath evaluation |
20. Operator Skill and Process Discipline
Electroplating is highly process-sensitive.
Experienced operators understand:
- chemistry trends,
- current distribution,
- contamination control,
- troubleshooting.
Many failures come from inconsistent procedures.
Example: High-Quality Nickel-Chrome Plating Process
Typical sequence:
Polishing
↓
Degreasing
↓
Alkaline Cleaning
↓
Acid Activation
↓
Copper Strike
↓
Nickel Plating
↓
Chrome Plating
↓
Rinsing
↓
Drying
↓
Inspection
Common Features of Excellent Electroplating
| Feature | Result |
|---|---|
| Smooth deposit | Attractive finish |
| Uniform thickness | Reliable protection |
| Strong adhesion | Long service life |
| Low porosity | Better corrosion resistance |
| Fine grain structure | Improved hardness |
| Low contamination | Fewer defects |
Most Important Factors (Priority Order)
The biggest quality factors are:
- Surface preparation
- Bath chemistry control
- Current density control
- Cleanliness
- Temperature stability
- Filtration and agitation
- Proper rinsing
Summary
High-quality electroplating requires:
- extremely clean surfaces,
- carefully controlled bath chemistry,
- correct current density,
- stable temperature,
- proper agitation,
- contamination prevention,
- accurate thickness control,
- and rigorous process monitoring.
The best electroplating operations maintain tight control over every stage — from cleaning and activation to plating, rinsing, and post-treatment.
Other courses:



