A hydrogen fuel cell is a device that generates electricity through a chemical reaction between hydrogen and oxygen.
It produces electric power, heat, and water, with no carbon emissions at the point of use.
Fuel cells are used in vehicles, backup power systems, and clean energy applications.
In this article:
- What is the hydrogen fuel cell?
- 1. What Makes a Hydrogen Fuel Cell Different
- 2. Core Components of a Hydrogen Fuel Cell
- 3. How a Hydrogen Fuel Cell Works (Step by Step)
- 4. Types of Hydrogen Fuel Cells (By Technology)
- 5. Efficiency and Energy Performance
- 6. Advantages of Hydrogen Fuel Cells
- 7. Limitations and Challenges
- 8. Hydrogen Fuel Cells vs Battery Electric Vehicles
- 9. Real-World Applications
- 10. The Big Picture
- Other courses:
What is the hydrogen fuel cell?
A hydrogen fuel cell is an electrochemical device that produces electricity by combining hydrogen and oxygen, with water and heat as the only byproducts. Unlike engines or turbines, it does not burn fuel—it converts chemical energy directly into electrical energy, making it highly efficient and clean at the point of use.
Below is a deep, structured explanation covering how it works, types, efficiency, advantages, limitations, and real-world uses.
1. What Makes a Hydrogen Fuel Cell Different
| Feature | Fuel Cell | Battery | Combustion Engine |
|---|---|---|---|
| Energy source | External hydrogen | Stored electricity | Burned fuel |
| Emissions | Water only | None (use phase) | CO₂, NOₓ |
| Efficiency | 50–60% (up to 85% with heat recovery) | ~90% | 20–30% |
| Refueling | Minutes | Hours | Minutes |
A fuel cell keeps producing electricity as long as hydrogen is supplied, unlike a battery that must be recharged.
2. Core Components of a Hydrogen Fuel Cell
Main parts
- Anode – where hydrogen enters
- Cathode – where oxygen enters
- Electrolyte – allows ions to pass, blocks electrons
- Catalyst – usually platinum, speeds reactions
- External circuit – electrons flow here → usable power
3. How a Hydrogen Fuel Cell Works (Step by Step)
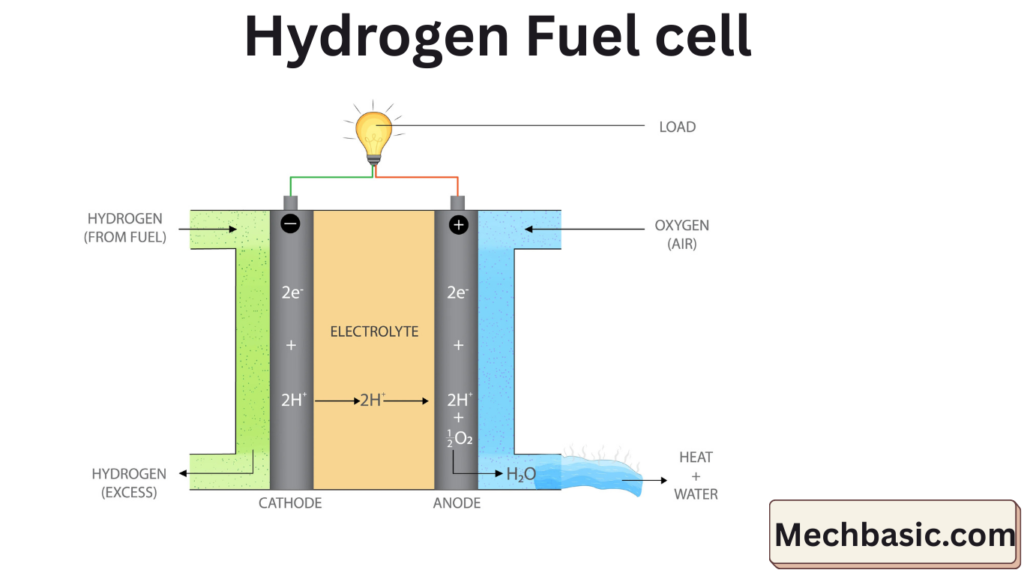
Step 1: Hydrogen splitting (Anode)
Hydrogen gas (H₂) enters the anode and is split by the catalyst:
H₂ → 2H⁺ + 2e⁻
- Protons (H⁺) move through the electrolyte
- Electrons (e⁻) are forced through an external circuit → electricity
Step 2: Electricity generation
- Electrons flowing through the circuit power:
- Motors
- Electronics
- Grids or buildings
Step 3: Water formation (Cathode)
At the cathode, oxygen reacts with protons and electrons:
½O₂ + 2H⁺ + 2e⁻ → H₂O
- Output: water vapor + heat
- No carbon emissions at the point of use
4. Types of Hydrogen Fuel Cells (By Technology)
1. Proton Exchange Membrane Fuel Cell (PEMFC)
- Low operating temperature (~80°C)
- Fast startup
- Used in cars, buses, trucks
- Sensitive to fuel purity
2. Solid Oxide Fuel Cell (SOFC)
- High temperature (600–1,000°C)
- Very efficient
- Used for stationary power
- Slow startup, long lifespan
3. Alkaline Fuel Cell (AFC)
- Used historically in space missions
- High efficiency
- Requires ultra-pure hydrogen
4. Molten Carbonate Fuel Cell (MCFC)
- Industrial-scale power
- Can use natural gas-derived hydrogen
- Large and complex
5. Efficiency and Energy Performance
- Electrical efficiency: 50–60%
- Combined heat & power (CHP): up to 85–90%
- Hydrogen energy density (by mass):
- ~120 MJ/kg (≈3× gasoline)
⚠️ By volume, hydrogen is low-density → storage is challenging.
6. Advantages of Hydrogen Fuel Cells
Environmental
- Zero tailpipe emissions
- Quiet operation
- No particulate pollution
Technical
- Fast refueling (3–5 minutes)
- Long driving range (500–700 km)
- Scales from watts to megawatts
Energy system benefits
- Enables renewable energy storage
- Supports energy security
- Works where batteries struggle (heavy transport, aviation, shipping)
7. Limitations and Challenges
Hydrogen production
- Most hydrogen today is made from natural gas (CO₂ emissions)
- Green hydrogen (from electrolysis) is still costly
Storage & transport
- Requires:
- High pressure (350–700 bar), or
- Cryogenic temperatures (−253°C)
Cost
- Platinum catalysts are expensive
- Infrastructure is limited
Energy losses
Electricity → hydrogen → electricity has lower round-trip efficiency than batteries
8. Hydrogen Fuel Cells vs Battery Electric Vehicles
| Aspect | Fuel Cell | Battery |
|---|---|---|
| Refueling time | Minutes | Hours |
| Range | Long | Medium |
| Efficiency | Lower | Higher |
| Infrastructure | Limited | Growing fast |
| Best for | Heavy-duty, long-haul | Cars, urban use |
👉 Fuel cells complement batteries, not replace them.
9. Real-World Applications
Transportation
- Cars, buses, trucks, trains
- Forklifts and port equipment
Stationary power
- Backup power for hospitals and data centers
- Distributed generation
Industrial & future uses
- Steelmaking
- Aviation (hybrid systems)
- Grid-scale energy storage
10. The Big Picture
Hydrogen fuel cells are not a silver bullet, but they are a critical piece of a low-carbon energy system, especially for:
- Heavy transport
- Long-duration energy storage
- Industries hard to electrify
Summary
A hydrogen fuel cell is a clean, efficient device that generates electricity by electrochemically combining hydrogen and oxygen, producing only water—offering major benefits for transportation and power where batteries fall short.
Other courses:



