Powder preparation in powder metallurgy involves producing fine metal powders from bulk metal.
Common methods include atomization, chemical reduction, electrolysis, and mechanical grinding.
The choice of method depends on the required particle size, shape, and purity.
In this article:
Powder preparation in Powder Metallurgy (PM) is the first and one of the most important stages, where metals are converted into fine powders suitable for compaction and sintering. The quality of powder directly controls the strength, density, and performance of the final product.
Powder Preparation in Powder Metallurgy
What is Powder Preparation?
It is the process of producing metal powders with controlled size, shape, purity, and composition for use in powder metallurgy.
Methods of Powder Preparation
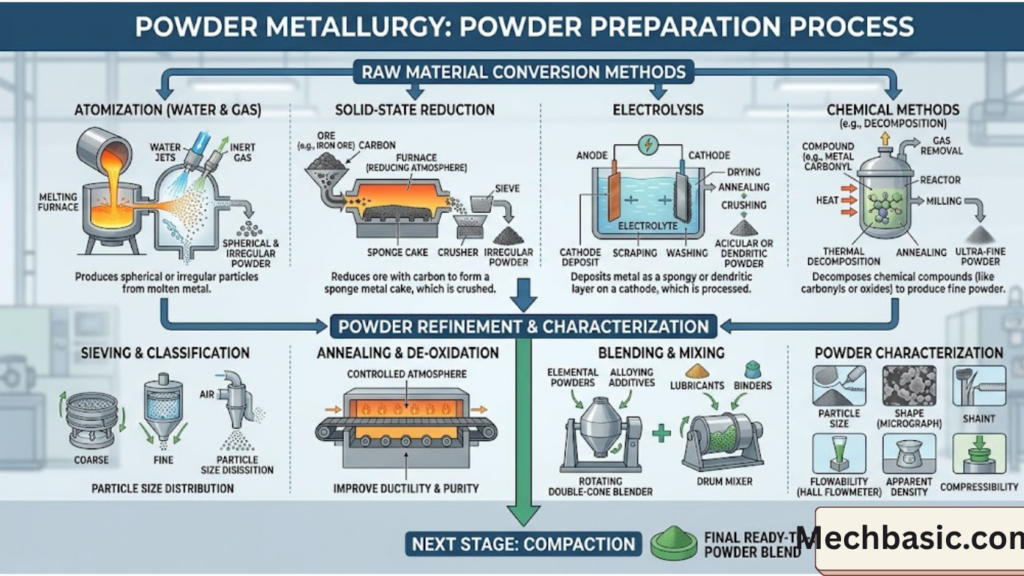
1. Atomization Method (Most Common)
Principle
Molten metal is broken into fine droplets, which solidify into powder.
Types of atomization:
(a) Gas Atomization
- High-pressure gas (argon, nitrogen) breaks molten metal stream
- Produces spherical particles
(b) Water Atomization
- High-pressure water jets used
- Produces irregular-shaped particles
Advantages
- High production rate
- Suitable for many metals and alloys
Applications
- Steel powders
- Aluminium powders
- Superalloys
2. Mechanical Method
Process
- Bulk metal is crushed, ground, or milled into powder
Equipment used
- Ball mills
- Hammer mills
- Attrition mills
Features
- Produces irregular, angular particles
- Suitable for brittle metals
Applications
- Tungsten powder
- Magnesium powders
3. Chemical Reduction Method
Principle
Metal oxides are reduced using gases like hydrogen or carbon monoxide.
Example
Iron oxide → Iron powder (using hydrogen)
Advantages
- High purity powders
- Fine particle size
Applications
- Iron powders (very common in PM)
4. Electrolytic Method
Process
- Metal is deposited from a solution using electric current
- Deposited metal is scraped and converted into powder
Features
- Very pure powders
- Dendritic (branch-like) structure
Applications
- Copper powder
- Iron powder
5. Thermal Decomposition Method
Principle
Metal compounds are heated until they break down into metal powder.
Example
Nickel carbonyl → Nickel powder + gas
Advantages
- Very fine, uniform powders
6. Carbonyl Process
Used for
- Nickel and iron powders
Process
- Metal reacts with carbon monoxide → forms volatile compound
- Then decomposed to pure metal powder
Applications
- High-purity nickel powders
7. Precipitation from Solution
Process
- Metal salts are chemically precipitated from solution
- Then reduced to powder
Features
- Very fine particles
- Controlled composition
Comparison Table
| Method | Powder Shape | Purity | Common Use |
|---|---|---|---|
| Atomization | Spherical/irregular | Medium | Most metals |
| Mechanical | Irregular | Medium | Brittle metals |
| Chemical Reduction | Fine powder | High | Iron |
| Electrolytic | Dendritic | Very high | Copper, iron |
| Carbonyl Process | Very fine | Very high | Ni, Fe |
| Thermal Decomposition | Fine | High | Nickel |
Key Factors in Powder Preparation
Good powder should have:
1. Particle Size
- Fine but controlled size improves compaction
2. Particle Shape
- Spherical → better flow
- Irregular → better bonding
3. Purity
- Impurities reduce strength
4. Flowability
- Powder should flow easily into dies
5. Compressibility
- Should compact easily under pressure
Summary:
👉 The most widely used method is atomization, because it:
- Works for many metals
- Produces good quality powders
- Is cost-effective for mass production
Conclusion:
Powder preparation in powder metallurgy involves producing metal powders using methods such as atomization, mechanical grinding, chemical reduction, electrolysis, and thermal decomposition. These methods control particle size, shape, and purity.
Other courses:



