Sintering is a heat treatment process used to bond powdered materials without fully melting them.
It strengthens the compacted powder by causing particles to fuse together at high temperature.
It is widely used in powder metallurgy to improve density and mechanical properties.
In this article:
Sintering (Powder Metallurgy)
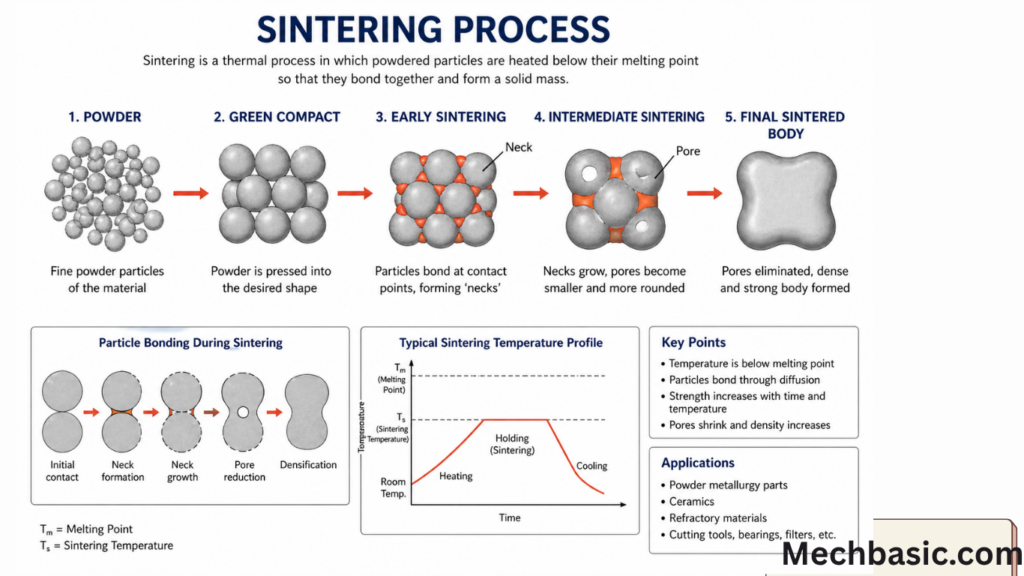
Sintering is the most important step in powder metallurgy, where a compacted powder part is heated below its melting point so that particles bond together and form a strong solid component.
Definition
👉 Sintering is a heat treatment process in which a pressed powder compact (green body) is heated below its melting point to cause diffusion bonding between particles, increasing strength and density.
Where Sintering Happens in PM Process
- Powder production
- Mixing/blending
- Compaction → (green compact)
- 🔥 Sintering
- Finishing operations
Step-by-Step Sintering Process
1. Heating Stage
- Compact is placed in a controlled furnace
- Heated to about:
- 0.6 to 0.9 of melting point (Tm)
- Atmosphere is controlled (to avoid oxidation)
2. Initial Bond Formation (Neck Formation)
- Powder particles touch each other
- Atoms start moving across boundaries
- Small “necks” form between particles
👉 This is the first stage of solid bonding
3. Neck Growth
- Continued diffusion enlarges the necks
- Particles become strongly attached
- Strength increases significantly
4. Densification
- Voids (pores) between particles shrink
- Material becomes more compact
- Density increases
5. Cooling Stage
- Controlled cooling in furnace or environment
- Prevents cracks and distortion
- Final strong solid part is obtained
Mechanism of Sintering
Sintering works mainly through:
Atomic Diffusion
- At high temperature, atoms move between particles
- Causes bonding without full melting
Types of diffusion involved:
- Surface diffusion
- Grain boundary diffusion
- Volume diffusion
Types of Sintering
1. Solid-State Sintering
- No liquid phase
- Most common method
👉 Used for iron, copper, steel parts
2. Liquid Phase Sintering
- Small amount of material melts
- Liquid helps bonding and densification
👉 Used in tungsten carbide tools
3. Activated Sintering
- Additives speed up diffusion
- Reduces required temperature/time
4. Hot Pressing / Hot Isostatic Pressing (HIP)
- Heat + pressure applied together
- Produces near-zero porosity
Factors Affecting Sintering
1. Temperature
- Higher temperature → better bonding
- Must remain below melting point
2. Time
- Longer time → better densification
3. Atmosphere
- Hydrogen / nitrogen / vacuum used
- Prevents oxidation
4. Particle Size
- Smaller particles → faster sintering
5. Pressure (in advanced methods)
- Improves density and strength
Effects of Sintering
Improves:
- Strength
- Hardness
- Density
- Wear resistance
- Structural integrity
Reduces:
- Porosity
- Weak particle boundaries
Sintering Stages Summary
| Stage | Process |
|---|---|
| 1 | Heating |
| 2 | Neck formation |
| 3 | Neck growth |
| 4 | Densification |
| 5 | Cooling |
Applications of Sintering
- Gears
- Bearings
- Cutting tools
- Electrical contacts
- Automotive components
Summary:
👉 Sintering is the heart of powder metallurgy because it converts a weak powder compact into a strong solid material through atomic diffusion (without melting).
Conclusion:
Sintering is the process of heating a compacted powder below its melting point to cause diffusion bonding between particles, resulting in increased strength, density, and structural integrity.
Other courses:



